Biomolecules
Biomolecules are molecules that occur naturally in living organisms. Biomolecules include macromolecules like proteins, carbohydrates, lipids, amino acids and nucleic acids. It also includes small molecules like primary and secondary metabolites and natural products. Biomolecules consist mainly of carbon and hydrogen with nitrogen, oxygen, sulphur and phosphorus. Biomolecules are very large molecules of many atoms which are covalently bound together. Carbohydrates serve as a major energy source, lipids contribute to energy storage and cell membrane formation, nucleic acids carry genetic information, and proteins perform diverse functions, including catalysing biochemical reactions and providing structural support.
Carbohydrates provide energy, lipids store energy and form membranes, proteins perform diverse functions, and nucleic acids carry genetic information.
Carbohydrates
Carbohydrates are the most abundant organic molecules in nature. They are primarily composed of the elements carbon, hydrogen and oxygen. The name carbohydrate literally means ‘hydrates of carbon’.
Classification of Carbohydrates
Carbohydrates are often referred to as saccharides (Greek: sakcharon–sugar). They are broadly classified into three major groups— monosaccharides, oligosaccharides and polysaccharides. This categorization is based on the number of sugar units.
Mono- and oligosaccharides are sweet to taste, crystalline in character and soluble in water, hence they are commonly known as sugars.
Monosaccharides
Monosaccharides (Greek: mono-one) are the simplest group of carbohydrates and are often referred to as simple sugars. They have the general formula Cn(H2O)n, and they cannot be further hydrolysed. The monosaccharides are divided into different categories, based on the functional group and the number of carbon atoms..
- Aldoses : When the functional group in monosaccharides is an aldehyde, they are known as aldoses e.g. glyceraldehyde, glucose.
- Ketoses : When the functional group is a keto C=O group, they are referred to as ketoses e.g. dihydroxyacetone, fructose.

Oligosaccharides
Oligosaccharides (Greek: oligo-few) contain 2-10 monosaccharide molecules held together by a glycosidic bond. Based on the number of monosaccharide units present, the oligosaccharides are further subdivided to disaccharides, trisaccharides etc.
Polysaccharides
Polysaccharides (Greek: poly-many) are polymers of monosaccharide units with high molecular weight (up to a million). They are usually tasteless (non-sugars) and form colloids with water. The polysaccharides are of two types – homopolysaccharides and heteropolysaccharides.
Starch is a homopolymer composed of D-glucose units held by D-glycosidic bonds. It is known as glucosan or glucan. Hyaluronic acid is a heteropolymer, composed of alternate units of D-glucuronic acid and N-acetyl D-glucosamine.
Biological Role of Carbohydrates
The biological role of carbohydrates encompasses several critical functions across organisms:
- Primary Energy Source: Glucose, a simple sugar, is essential for producing ATP, the cell's energy.
- Energy Storage: Plants and animals store excess glucose as starch and glycogen, respectively, for future energy needs.
- Structural Support: Cellulose in plant cell walls and chitin in fungal cell walls and arthropod exoskeletons provide structural integrity.
- Cell Communication: Glycoproteins and glycolipids on cell surfaces are key in cell recognition and signaling.
- Immune Function: Carbohydrates on pathogen surfaces trigger to initiate immune responses.
- Digestive Health: Dietary fibers aid in digestion and maintaining gut health.
- Genetic Information: Ribose and deoxyribose sugars are vital components of RNA and DNA.
Lipids
Lipids may be regarded as organic substances relatively insoluble in water, soluble in organic solvents (alcohol, ether etc.). Lipids are organic compounds containing long hydrocarbon chains (from fatty acids) along with functional groups such as carboxyl, alcohol, and sometimes phosphate groups.
Lipids are broadly classified into simple, complex and derived lipids.Classification of Lipids
Simple lipids : Esters of fatty acids with alcohols.
- Fats and oils (triacylglycerols) : These are esters of fatty acids with glycerol. The difference between fat and oil is only physical. Thus, oil is a liquid while fat is a solid at room temperature.
- Waxes : Esters of fatty acids (usually long chain) with alcohols other than glycerol.
- Phospholipids : They contain phosphoric acid and frequently a nitrogenous base. This is in addition to alcohol and fatty acids.
- Glycerophospholipids : These phospholipids contain glycerol as the alcohol.
- Sphingophospholipids : Sphingosine is the alcohol in this group of phospholipids.
- Glycolipids : These lipids contain a fatty acid, carbohydrate and nitrogenous base.
Derived Proteins: Derived lipids are those lipids that are formed when simple or complex lipids are broken down (hydrolyzed). Example: mono- and diacylglycerols (derived from the partial hydrolysis of triglycerides), lipid (fat)soluble vitamins (Vitamin A, D, E and K), steroid hormones, hydrocarbons and ketone bodies.
Biological Role of Lipids
Lipids perform several important functions;- Energy storage: They are the concentrated fuel reserve of the body (triacylglycerols).
- Membrane structure: Lipids are the constituents of membrane structure and regulate the membrane permeability (phospholipids and cholesterol).
- Vitamin source: They serve as a source of fat soluble vitamins (A, D, E and K).
- Hormone regulation: Lipids are important as cellular metabolic regulators (steroid hormones and prostaglandins).
- Protection: Lipids protect the internal organs, serve as insulating materials and give shape and smooth appearance to the body.
Amino Acids
Amino acids are the basic building blocks of proteins, each consisting of an amino group (–NH₂), a carboxyl group (–COOH), a hydrogen atom, and a variable side chain (R-group) attached to a central carbon.
The R-group determines their chemical properties and function. About 20 standard amino acids combine in different sequences to form proteins in the body.
Classification of Amino acids
Amino acid classification based on the structure.| Type | Example |
|---|---|
| Amino acids with aliphatic side chains | Glycine, Alaine, Leucine, isoleucine |
| Amino acids containing hydroxyl (—OH) groups | Threonine, Serine, Threonine |
| Sulphur containing amino acids | Cysteine, Methionine |
| Acidic amino acids and their amides | Asparagine, Aspartic acid |
| Basic amino acids | Lysne, Arginine |
| Aromatic amino acids | Tyrosine, Tryptophane |
Classification of amino acids based on polarity
Amino acids are classified on the basis of the polarity of their side chain (R-group) into these main groups:| Type | Example |
|---|---|
| Nonpolar (Hydrophobic) Amino Acids | Glycine, Alanine, Phenylalanine, Valine, Proline |
| Polar amino acids with no charge on ‘R’ group | Serine, Threonine, Asparagine, Tyrosine |
| Polar amino acids with positive ‘R’ group | Lysine, Arginine, Histidine |
| Polar amino acids with negative ‘R’ group : | Aspartic acid, Glutamic acid |
Nutritional classification of amino acids
Based on the nutritional requirements, amino acids are grouped into two classes—essential and nonessential:- Essential or indispensable amino acids : The amino acids which cannot be synthesized by the body and, therefore,
need to be supplied through the diet are called essential amino acids.
Non-essential or dispensable amino acids :The body can synthesize about 10 amino acids to meet the biological needs, hence they need not be consumed in the diet.
| Essential Amino acids | Non-essential Amino acids |
|---|---|
| Arginine, Valine, Histidine, Isoleucine, Leucine, Lysine, Methionine, Phenylalanine, Threonine, Tryptophan. | Glycine, Alanine, Serine, Cysteine, Aspartate, Asparagine, Glutamate, Glutamine, Tyrosine and Proline. |
Proteins
Proteins are large biological macromolecules made up of amino acids linked by peptide bonds. They play essential roles in the body, including catalysis (enzymes), structural support (collagen), transport (hemoglobin), and regulation (hormones). Proteins are highly specific in function due to their unique three-dimensional structure. They are composed mainly of carbon, hydrogen, oxygen, nitrogen, and sometimes sulfur. The sequence of amino acids determines the protein’s properties and function.
Protein structure is organized into four levels. The primary structure is the linear sequence of amino acids. The secondary structure includes local folding patterns like α-helix and β-sheet formed by hydrogen bonding. The tertiary structure is the overall 3D shape formed by interactions among side chains. The quaternary structure refers to the association of two or more polypeptide chains into a functional protein.Classification of Proteins
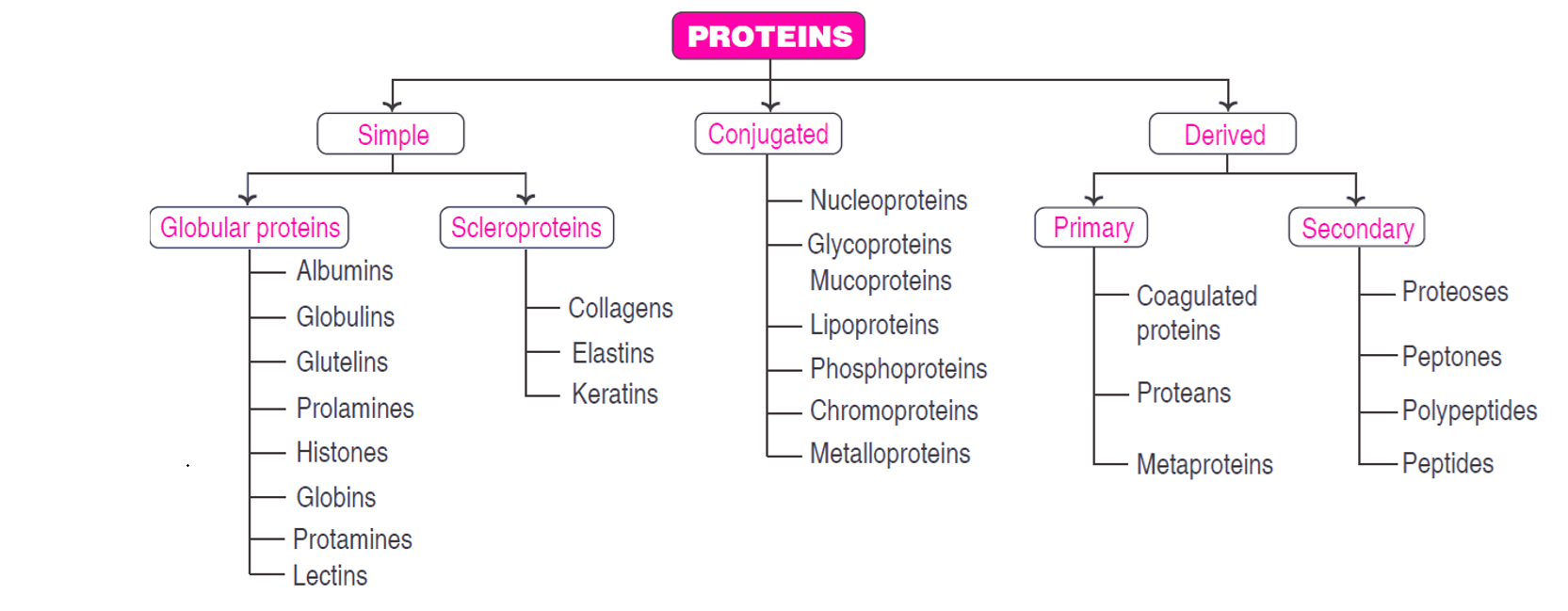
Classification of Proteins based on their functions
| Category | Example |
|---|---|
| Structural proteins | Keratin of hair and nails, collagen of bone |
| Enzymes or catalytic proteins | Amylase, Pepsin, Urease |
| Transport proteins | Hemoglobin, Serum albmin |
| Hormonal proteins | Various hormone |
| Contractile proteins | Actin, Myosin |
| Storage proteins | Ovalbumin, Glutelin |
| Genetic proteins | Nucleoproteins |
| Défense proteins | Antibodies |
Biological Role of Proteins
- Structural Function: Proteins form the structural components of cells and tissues. They provide strength and support to various body parts such as skin, bone, cartilage, hair, and nails. Examples include Collagen and Keratin.
- Enzymatic Function: Proteins act as enzymes and catalyze biochemical reactions in the body. These reactions are essential for digestion, metabolism, and energy production. Examples include Amylase and Pepsin.
- Transport Function: Proteins help in the transport of various substances across the body. They carry oxygen, nutrients, and metabolites. For example, Hemoglobin transports oxygen, while Albumin carries various molecules in blood.
- Protective Function: Proteins play a crucial role in the defense mechanism of the body. They form antibodies that protect against infections and diseases. Example: Antibodies.
- Hormonal Function: Some proteins function as hormones and regulate various physiological activities. These hormones control growth, metabolism, and homeostasis. Examples include Insulin and Growth hormone.
- Contractile Function: Proteins are responsible for muscle contraction and movement. They help in locomotion and mechanical work in the body. Examples include Actin and Myosin.
- Regulatory Function: Some proteins function as hormones and regulate various physiological activities. These hormones control growth, metabolism, and homeostasis. Examples include Insulin and Growth hormone.
- Energy Source: Some proteins function as hormones and regulate various physiological activities. These hormones control growth, metabolism, and homeostasis. Examples include Insulin and Growth hormone.
- Storage Function: Some proteins store essential substances for future use. For example, Ferritin stores iron in the body.
Nucleic Acids
Nucleic acids are the polymers of nucleotides. These nucleotides are attached to each other by 3’ and 5’ phosphate bridges. Nucleotides are composed of a nitrogenous base, a pentose sugar and a phosphate.
Nucleic acids refer to the genetic material found in the cell that carries all the hereditary information from parents to progeny.There are two types of nucleic acids, namely deoxyribonucleic acid (DNA) and ribonucleic acid (RNA).
in DNA, the sugar is 2-deoxyribose, which lacks an oxygen atom at the 2′ position compared to ribose, whereas In RNA, the sugar is ribose.DNA
- DNA (Deoxyribonucleic Acid) is the hereditary material present in almost all living organisms. It carries genetic information that controls growth, development, and reproduction.
- Structurally, DNA consists of two long strands forming a double helix.
- Each strand is made up of repeating units called nucleotides.
- A nucleotide contains a sugar (deoxyribose), a phosphate group, and a nitrogenous base.
- The four bases in DNA are adenine (A), thymine (T), cytosine (C), and guanine (G).
- These bases pair specifically (A with T, and C with G) through hydrogen bonds.
- DNA is mainly located in the nucleus of eukaryotic cells and directs protein synthesis.

RNA
- RNA (Ribonucleic Acid) is a single-stranded nucleic acid involved in protein synthesis. It is composed of ribose sugar, phosphate groups, and nitrogenous bases (adenine, uracil, cytosine, and guanine). Unlike DNA, RNA contains uracil instead of thymine.
- RNA is mainly found in the cytoplasm and nucleolus of the cell.
- There are three major types: mRNA (messenger RNA), tRNA (transfer RNA), and rRNA (ribosomal RNA).
- mRNA carries genetic information from DNA to ribosomes for protein synthesis.
- tRNA and rRNA help in assembling amino acids into proteins during translation.
Biological Role of Nucleic acids
- Storage of Genetic Information: DNA stores hereditary information in the form of genes.
- Transmission of Genetic Information: DNA replicates during cell division and transfer the genetic information from parent to offspring.
- Protein Synthesis: Nucleic acids (DNA and RNA) involves in the process of proteins synthesis.
- Regulation of Cellular Activities: Genes regulate enzyme production and control metabolism, growth, and differentiation of cells,
- Catalyzing biochemical reactions: (in the case of some RNA molecules).